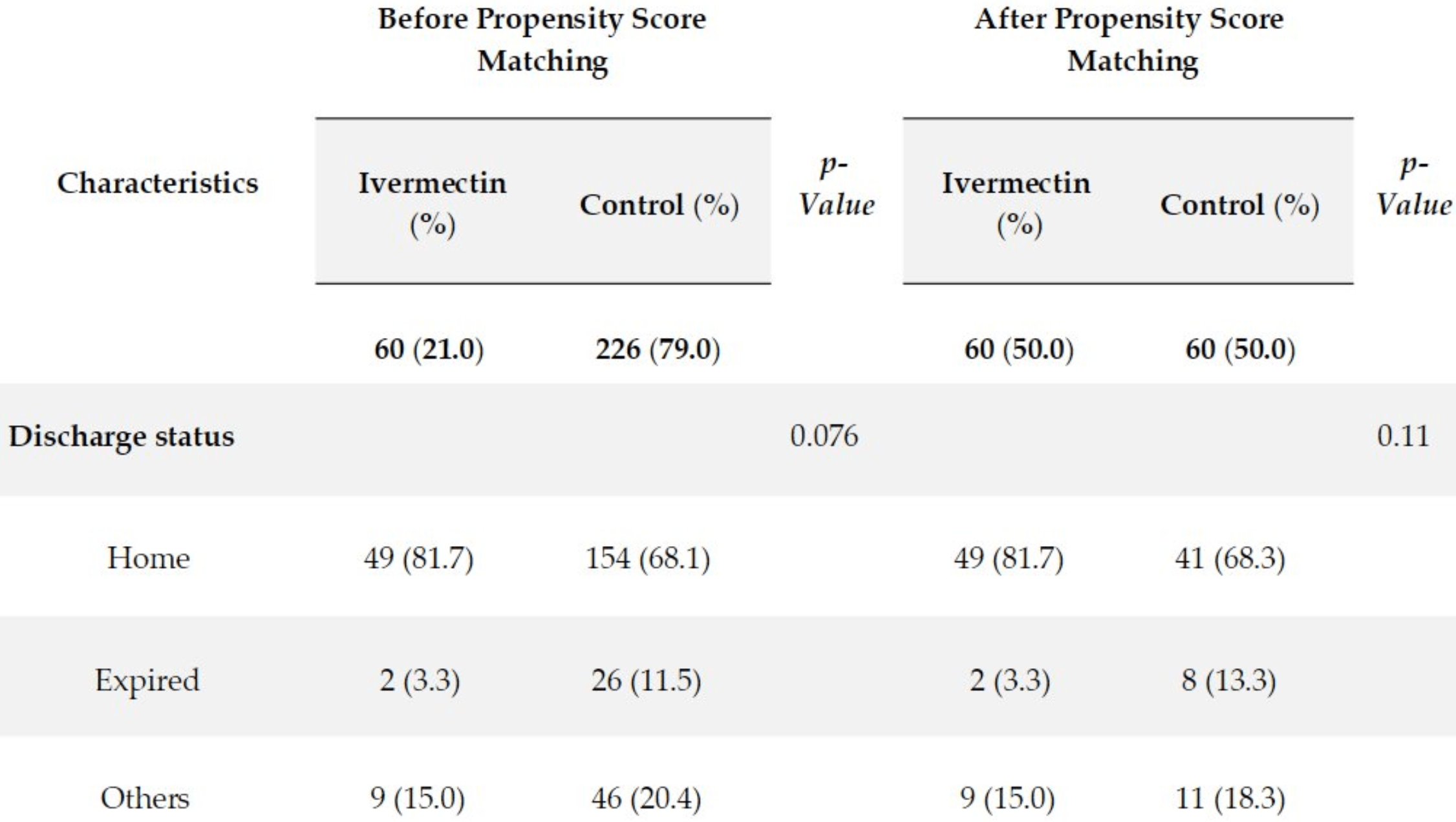
Small prospective PSM study in the USA, showing 75% lower mortality with ivermectin treatment, without reaching statistical significance, significantly shorter ventilation and ICU time, and longer hospitalization time.
Authors leave the statistically significant improvements in ventilation and ICU time out of the abtract and conclusions, and incorrectly state that there were no differences in other outcomes (there were no statistically significant differences)
nature.com. Authors are ambiguous on the primary outcome, referring to the primary mortality outcome in one case, and "clinical outcomes, measured by the rate of intubation, length of hospital stay, and mechanical ventilation duration" in another case.
The longer hospitalization time may be partially due to the greater mortality in the control group.
49 studies are RCTs, which show efficacy with
p=0.00000038.
|
risk of death, 75.0% lower, RR 0.25, p = 0.09, treatment 2 of 60 (3.3%), control 8 of 60 (13.3%), NNT 10.0, PSM.
|
|
risk of mechanical ventilation, 12.6% lower, RR 0.87, p = 0.20, treatment 3 of 60 (5.0%), control 2 of 60 (3.3%), adjusted per study, odds ratio converted to relative risk, propensity score matching, multivariable.
|
|
ventilation time, 83.3% lower, relative time 0.17, p = 0.002, treatment 60, control 60.
|
|
risk of ICU admission, 48.7% lower, RR 0.51, p = 0.42, treatment 6 of 60 (10.0%), control 3 of 60 (5.0%), adjusted per study, odds ratio converted to relative risk, propensity score matching, multivariable.
|
|
ICU time, 70.6% lower, relative time 0.29, p < 0.001, treatment 60, control 60.
|
|
hospitalization time, 9.0% higher, relative time 1.09, p = 0.09, treatment 60, control 60, adjusted per study, propensity score matching, multivariable.
|
|
Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates
|
Ozer et al., 23 Nov 2021, prospective, USA, peer-reviewed, 12 authors, dosage 200μg/kg days 1, 3.
Effectiveness and safety of Ivermectin in COVID‐19 patients: A prospective study at a safety‐net hospital
M.D. Muhammet Ozer, Suleyman Yasin Goksu, Reena Conception, MD Esad Ulker, Rodolfo Magallanes Balderas, MD Mohammed Mahdi, MD Zulfiya Manning, PharmD, BCPS, BCIDP Kim To, Muhammad Effendi, MD Rajashree Anandakrishnan, MD Marc Whitman, MD Manish Gugnani
Journal of Medical Virology, doi:10.1002/jmv.27469
Background: Ivermectin has been found to inhibit severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) replication in vitro. It is unknown whether this inhibition of SARS-CoV-2 replication correlates with improved clinical outcomes. Objective: To assess the effectiveness and safety of ivermectin in hospitalized patients with COVID-19. Methods and Materials: A total of 286 patients with COVID-19 were included in the study. Univariate analysis of the primary mortality outcome and comparisons between treatment groups were determined. Logistic regression and propensity score matching (PSM) was used to adjust for confounders. Results: Patients in the ivermectin group received 2 doses of ivermectin at 200 μg/kg in addition to usual clinical care on hospital days 1 and 3. The ivermectin group had a
References
Ahmed, A five-day course of ivermectin for the treatment of COVID-19 may reduce the duration of illness, International Journal of Infectious Diseases
Boldescu, Broad-spectrum agents for flaviviral infections: dengue, Zika and beyond, Nature Reviews Drug Discovery
Caly, The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Antiviral research
Camprubí, Lack of efficacy of standard doses of ivermectin in severe COVID-19 patients, Plos one
Eweas, Alhossary, Abdel-Moneim, Molecular docking reveals Ivermectin and Remdesivir as potential repurposed drugs against SARS-CoV-2, Frontiers in microbiology
Kory, Review of the Emerging Evidence Demonstrating the Efficacy of Ivermectin in the Prophylaxis and Treatment of COVID-19
Lehrer, Rheinstein, Ivermectin docks to the SARS-CoV-2 spike receptor-binding domain attached to ACE2, vivo
Muñoz, Safety and pharmacokinetic profile of fixed-dose ivermectin with an innovative 18mg tablet in healthy adult volunteers, PLoS neglected tropical diseases
Navarro, Safety of high-dose ivermectin: a systematic review and metaanalysis, Journal of Antimicrobial Chemotherapy
Rajter, Use of ivermectin is associated with lower mortality in hospitalized patients with Coronavirus Disease 2019: The ivermectin in COVID nineteen study, Chest
Rowland, Intracellular localization of the severe acute respiratory syndrome coronavirus nucleocapsid protein: absence of nucleolar accumulation during infection and after expression as a recombinant protein in vero cells, Journal of virology
Smit, Human direct skin feeding versus membrane feeding to assess the mosquitocidal efficacy of high-dose Ivermectin (IVERMAL Trial), Clinical Infectious Diseases
Timani, Nuclear/nucleolar localization properties of C-terminal nucleocapsid protein of SARS coronavirus, Virus research
Wagstaff, Ivermectin is a specific inhibitor of importin α/β-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus, Biochemical Journal
Wimmersberger, Efficacy and safety of ivermectin against Trichuris trichiura in preschool-aged and school-aged children: a randomized controlled dose-finding trial, Clinical Infectious Diseases
Yang, The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer, Antiviral research